2D and 3D Precipitation of a gas phase in a liquide phase
2D and 3D Precipitation of a gas phase in a liquide phase
This test case derived by [1] in 2D and [2] in 3D describes the precipitation of a pure gas phase into a liquid phase.
The study focuses here on the diffusion of a single chemical species, denoted \(A\). The second species composing the solution also diffuses, and the magnitude of its flux is equal to that of species \(A\).
We consider a box of side length \(L = 30 \times L_{\text{ref}}\) where \(L_{\text{ref}}\) represent the diameter of the pure gas phase. A composite mesh is employed, consisting of two distinct regions:
(i) The first region, centered on the gas phase, is composed of uniform cells. It extends up to a distance \(l_u = 4 \times L_{\text{ref}}\) from the center, in order to accurately capture the local dissolution phenomena. This zone is discretized with a fine resolution corresponding to 50 cells per diameter \(D\).
(ii) The second region covers the remainder of the domain. It uses a non-uniform mesh in which the spatial step increases exponentially as the distance from the bubble increases. The objective is to limit the total number of cells while maintaining sufficient resolution in the vicinity of the bubble.
The initial radius of the sphere is \(R = 5\times 10^{-3}~\text{m}\).
The properties of the system are:
| phases | \(\rho~\left[\text{kg}.\text{m}^{-3}\right]\) | \(\mu~\left[\text{N.s}.\text{m}^{-2}\right]\) |
|---|---|---|
| liquid | 1000 | \(1.05\times 10^{-3}\) |
| gas | 1 | \(1.46\times 10^{-5}\) |
The simulation is performed with a henry's coefficient \(H=0.2\) for a diffusion coefficient: \(D_{A,l} = 2 \times 10^{-5}\,\mathrm{m^2\,s^{-1}}\). Different configurations are tested where the initial concentration of the solution varies from \(0.4\) to \(0.8~\text{kg}.\text{m}^{-3}\).
Neumann conditions are applied on all the boundaries. For the 3D case, we use axisymmetric coordinates.
The initial concentration field, corresponding to the initial radius \(R\) is computed by using the theoretical solution derived by [1].
The results for the shifted method are presented in the following figures. Figure 1 show the results obtained for the 2D case and Figure 2 for the 3D axisymmetric case. Figure X.1 presents a comparison between the interface displacement kinetics obtained numerically and those predicted analytically. Figure X.2 illustrates the comparison of the concentration profiles for the fastest kinetics (i.e., for \(\mathrm{Sc} = 1.05\) and initial concentration equal to \(0.8~\text{kg}.\text{m}^{-3}\)). ).
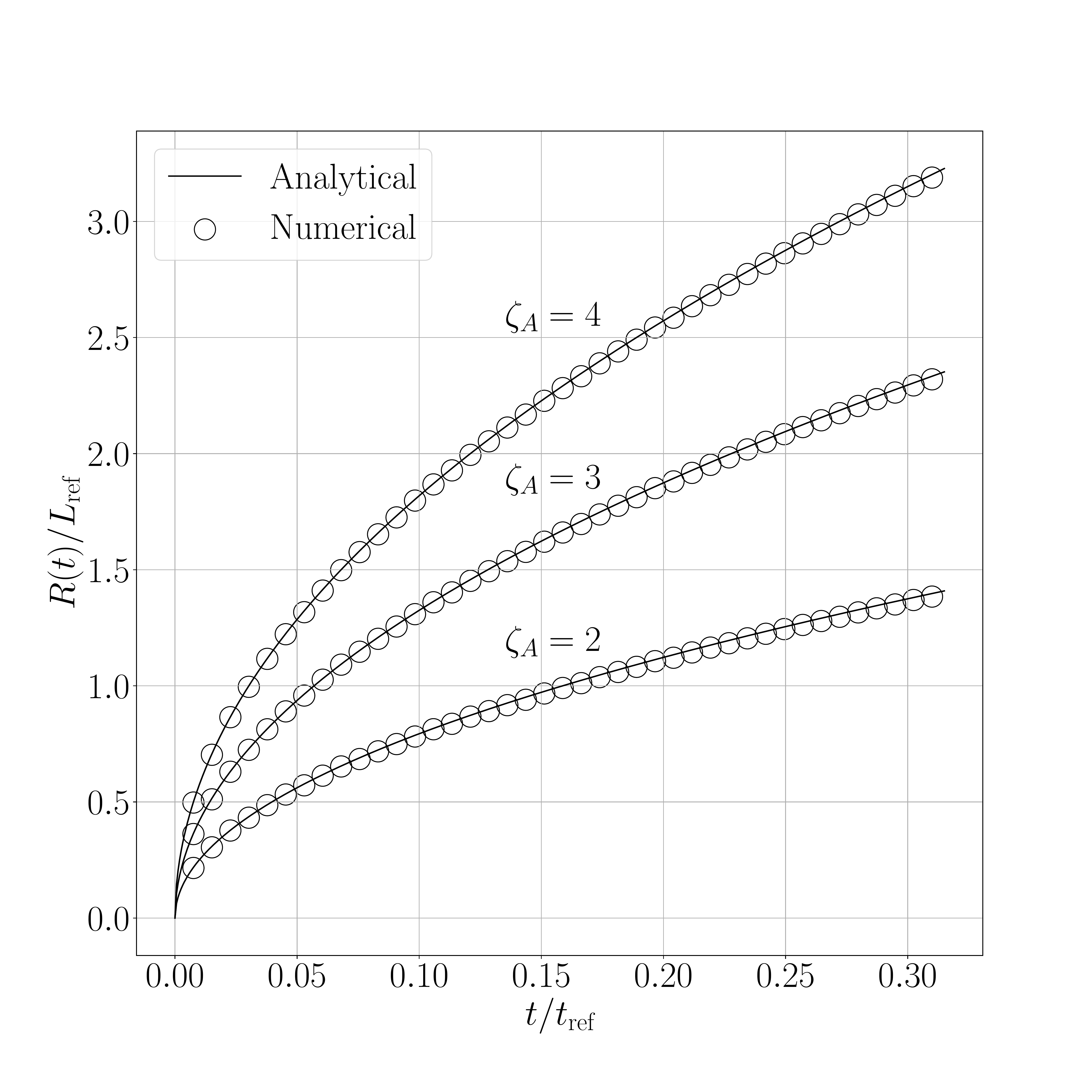
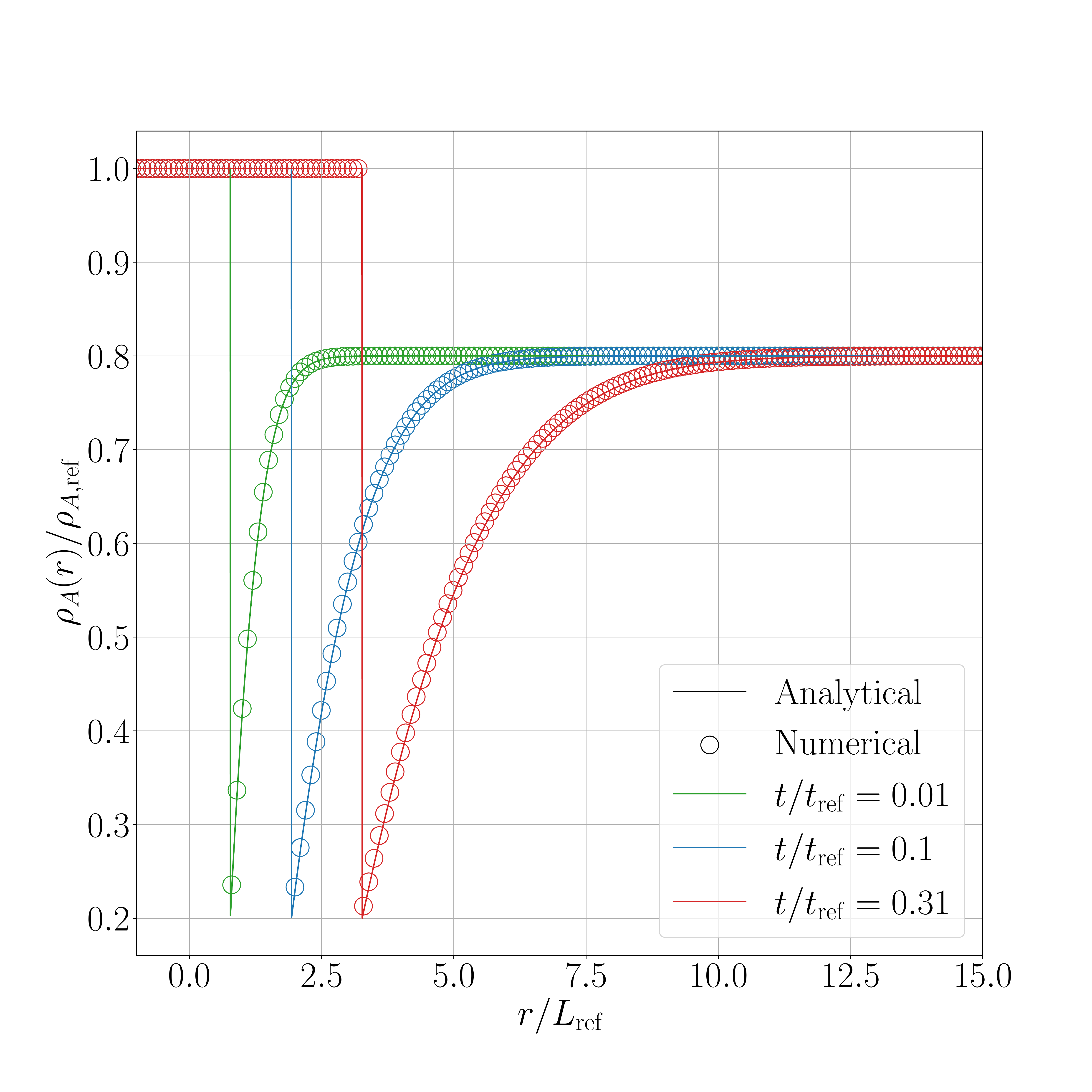
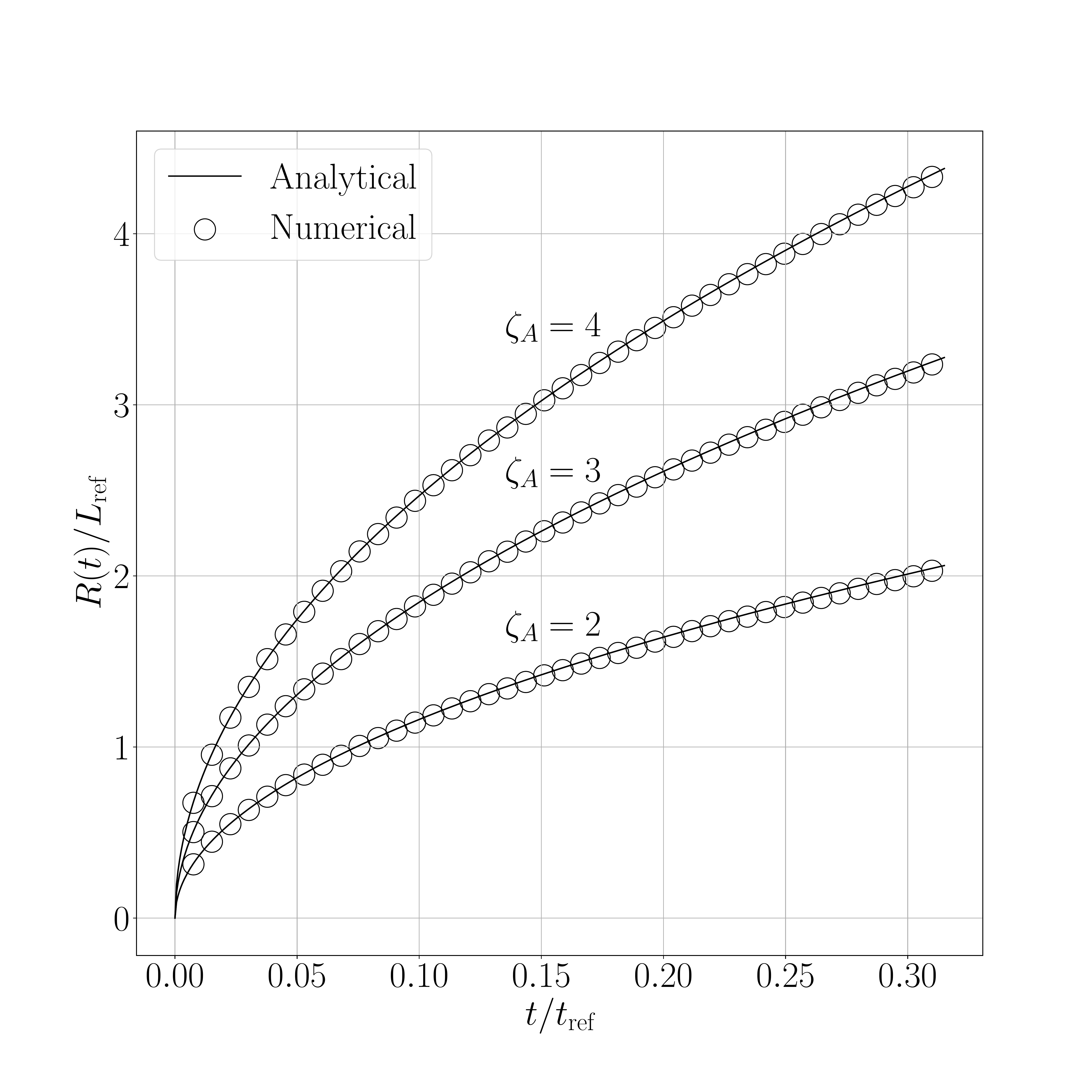
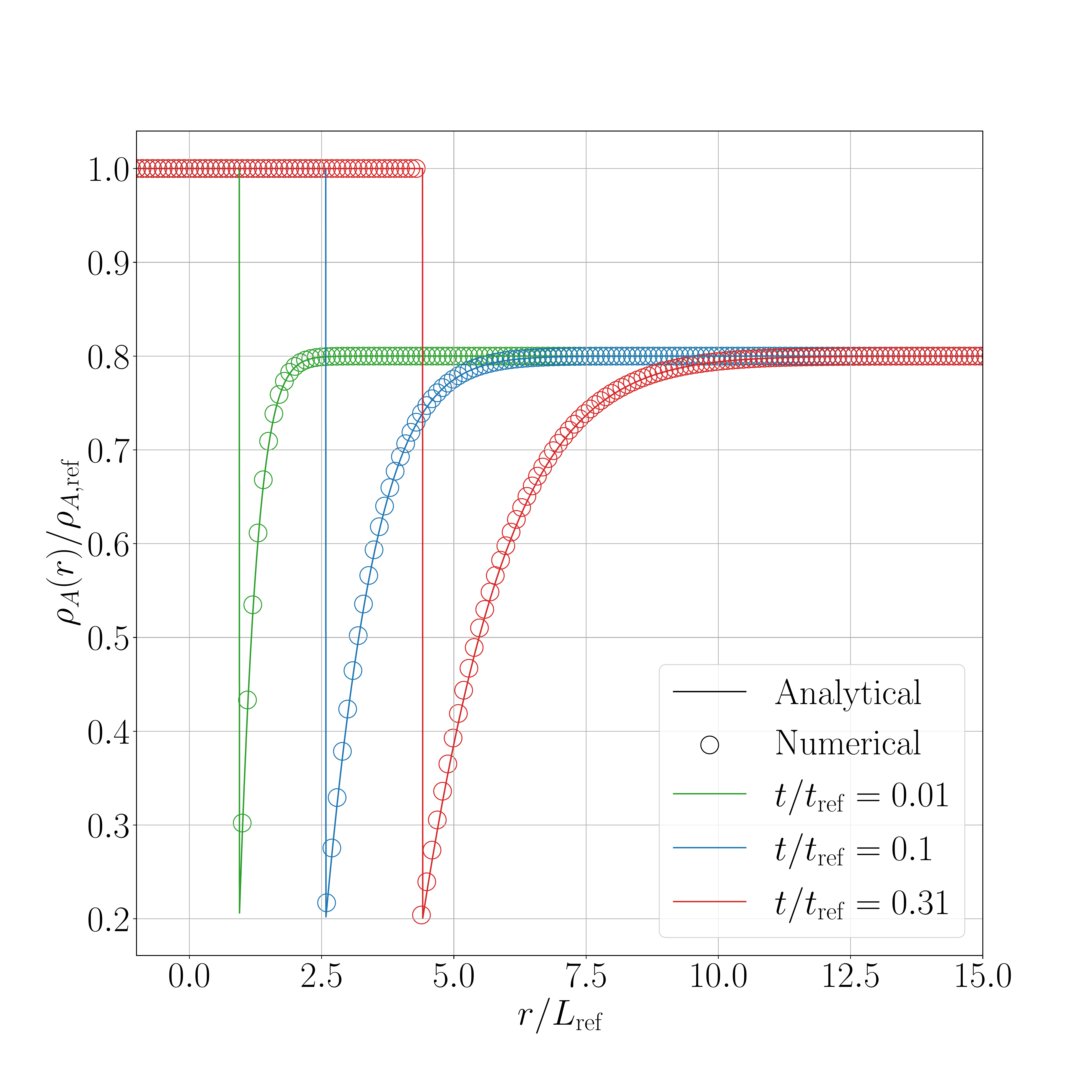
The obtained results show a good agreement between numerical and analytical solutions.
[1] Palka, C., Bordère, S., Lemoine, A., Glockner, S., & Erriguible, A. (2025). Simulation of precipitation/dissolution phenomena with large phase volume change using single-field C-CST model. Chemical Engineering Science, 317, 122074. https://doi.org/https://doi.org/10.1016/j.ces.2025.122074 [2] Scriven, L. (1959). On the dynamics of phase growth. Chemical Engineering Science, 10(1),1-13. https://doi.org/https://doi.org/10.1016/0009-2509(59)80019-1