1D Precipitation of a gas phase in a liquide phase
1D Precipitation of a gas phase in a liquide phase
This test case derived by [1] describes a flat interface separating a liquid phase from a gas phase. The pure gas phase precipitate into the liquid phase implying a flow in the surrounding fluid
This validation aims to confirm several key aspects: (i) the calculation of interfacial mass transfer, (ii) the volume variation of the gas phase, (iii) the variation of the mass fraction of chemical species during their transfer and its impact on the interface movement, and (iv) the accurate capture of the discontinuity of the concentration field across the interface.
The study focuses here on the diffusion of a single chemical species, denoted \(A\). The second species composing the solution also diffuses, and the magnitude of its flux is equal to that of species \(A\).
The study domain considered is a rectangle of arbitrary height and total length \(L = 10 \times L_{\text{ref}}\), where the characteristic length is set to \(L_{\text{ref}} = 1 \times 10^{-2}\,\mathrm{m}\).
The initial configuration places the interface between the phases at \(l_I(t=0) = 0\), thus defining a spatial distribution in which the gas phase occupies the left part of the domain \(-L/2 \leq x \leq l_I\), while the liquid phase is located in the right part \(l_I \leq x \leq L/2\).
The spatial discretization is based on a structured mesh comprising 1600 uniform cells in the \(x\)-direction. The number of cells in the \(y\)-direction remains arbitrary due to the one-dimensional nature of the problem.
The properties of the system are:
| phases | \(\rho~\left[\text{kg}.\text{m}^{-3}\right]\) | \(\mu~\left[\text{N.s}.\text{m}^{-2}\right]\) |
|---|---|---|
| liquid | 1000 | \(1.05\times 10^{-3}\) |
| gas | 1 | \(1.46\times 10^{-5}\) |
The simulation is performed with a henry's coefficient \(H=0.2\) and for a diffusion coefficient: \(D_{A,l} = 2 \times 10^{-5}\,\mathrm{m^2\,s^{-1}}\). Different configurations are tested where the initial concentration of the solution varies from \(0.4\) to \(0.8~\text{kg}.\text{m}^{-3}\).
A Neumann-type condition (outflow condition) is imposed at the right boundary of the domain at \(x = L/2\), while symmetry conditions are applied on the other boundaries.
The results for the shifted method are presented in the following figures. Figure 1 presents a comparison between the interface displacement kinetics obtained numerically and those predicted analytically. Figure 2 illustrates the comparison of the concentration profiles for the fastest kinetics (i.e., for an initial concentration inside the liquid phase equal to \(0.8~\text{kg}.\text{m}^{-3}\)).
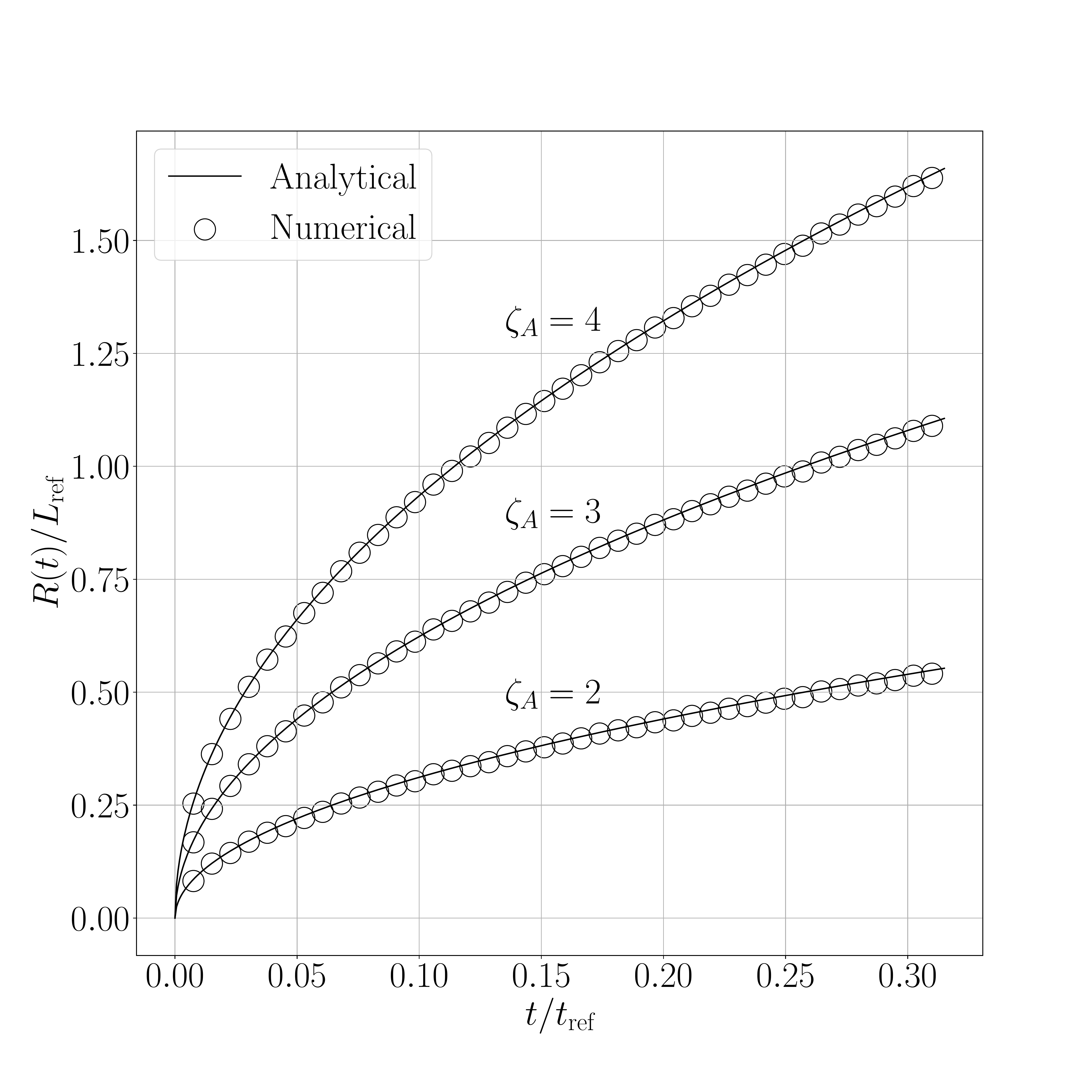
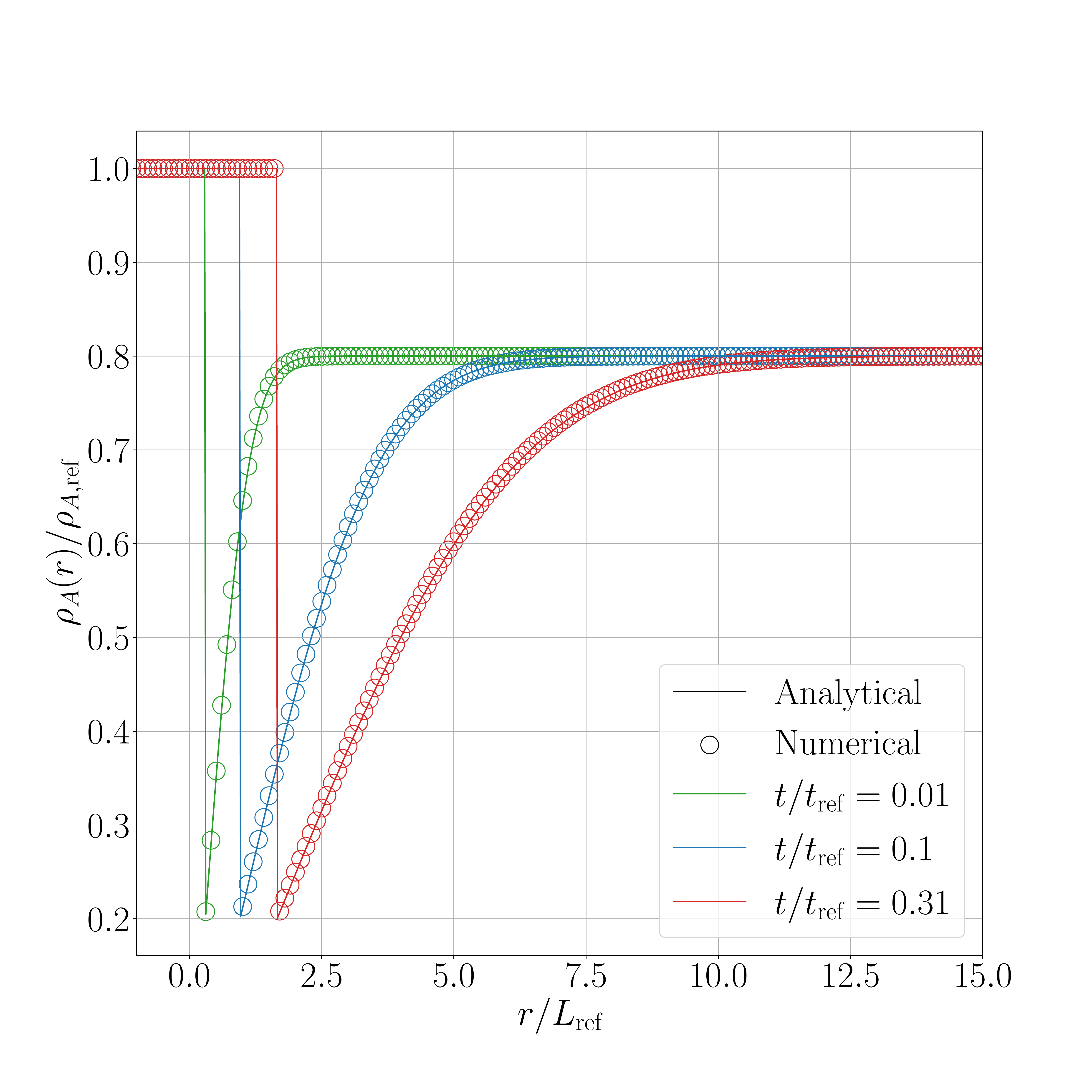
The obtained results show an excellent agreement between numerical and analytical solutions, regardless the value of the supersaturation.
[1] Palka, C., Bordère, S., Lemoine, A., Glockner, S., & Erriguible, A. (2025). Simulation of precipitation/dissolution phenomena with large phase volume change using single-field C-CST model. Chemical Engineering Science, 317, 122074. https://doi.org/https://doi.org/10.1016/j.ces.2025.122074